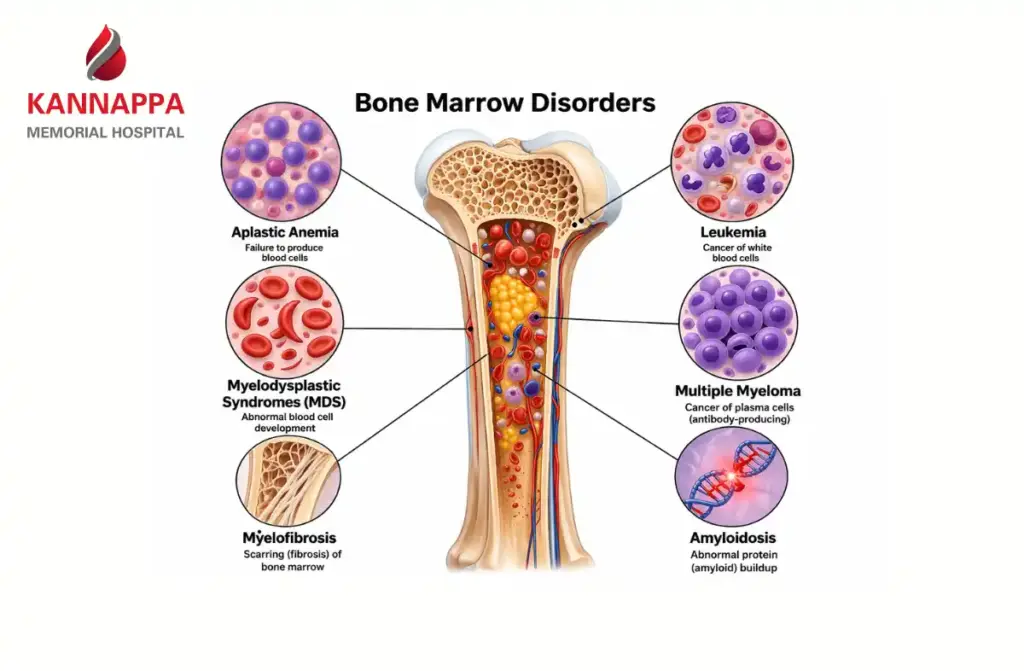
Bone marrow is the lifeline of the human hematopoietic system the complex biological network that continuously produces blood cells necessary for life. Disorders affecting the marrow, including bone marrow disorders, can lead to potentially life‑threatening conditions with far‑reaching impacts on immunity, oxygen transport, and blood clotting. At institutions like Kannappa Memorial Hospital, advanced diagnosis and multidisciplinary care are critical to properly manage people with these complex conditions.
This guide provides an in‑depth look at the structure and function of bone marrow, causes of bone marrow disorder and marrow failure, common diseases, clinical signs and symptoms, diagnostic approaches, treatment options, prognosis, and practical tips for living with marrow dysfunction.Understanding bone marrow disorders is essential because early recognition and treatment can dramatically improve outcomes and quality of life.
Bone Marrow and Stem Cells
What is Bone Marrow?
Bone marrow is a soft, spongy tissue found in the center of bones, particularly the pelvis, sternum, ribs, vertebrae, and long bones like the femur. Its primary role is to produce blood cells a process called hematopoiesis.
Types of Bone Marrow Cells
Within this tissue are multiple cell types, each performing vital roles:
- Red Blood Cells (RBCs): RBCs transport oxygen from the lungs to tissues and return carbon dioxide back for exhalation. They are crucial for energy metabolism and overall vitality.
- White Blood Cells (WBCs): White blood cells (WBCs) are essential to the immune system, protecting the body by fighting infections and harmful pathogens. They include neutrophils, lymphocytes, monocytes, eosinophils, and basophils each with a specific role in immune defense.
- Platelets (Thrombocytes): Platelets are responsible for blood clotting. They help stop bleeding by forming clots at injury sites, preventing excessive blood loss.
Role of Stem Cells in Marrow Function
At the heart of marrow activity are hematopoietic stem cells (HSCs) primitive, self‑renewing cells capable of differentiating into all blood cell types. HSCs maintain lifelong blood cell production through carefully regulated division and differentiation.
Inherited and Acquired Marrow Dysfunction
Bone marrow failure can be inherited (genetic) or acquired.
- Inherited: Conditions are present at birth due to mutations in key genes regulating blood formation.
- Acquired: Dysfunction develops later in life because of environmental exposures, autoimmune mechanisms, infections, or unknown triggers.
Differentiating these is essential for proper treatment planning and genetic counseling something expert teams at Kannappa Memorial Hospital specialize in.
Causes of Bone Marrow Failure
Bone marrow failure arises when the marrow doesn’t produce sufficient blood cells. Its causes range from genetic syndromes to environmental insults.
Genetic / Congenital Syndromes
These are rare but critical to identify:
- Fanconi Anemia — a DNA repair defect leading to progressive marrow failure.
- Diamond‑Blackfan Anemia — selectively affects red cell production.
- Shwachman‑Diamond Syndrome — affects marrow and pancreatic function.
These syndromes often require lifelong monitoring and specialized care, including possible stem cell transplantation.
Acquired Causes
- Aplastic Anemia: Aplastic anemia is a condition where the marrow fails to produce any type of blood cell. In many cases, it results from autoimmune destruction of stem cells.
- Myelodysplastic Syndromes (MDS): In MDS, cells are produced but are abnormal and ineffective, leading to cytopenias (low blood counts). MDS carries a risk of progression to acute leukemia.
- Myeloproliferative Disorders (MPD): These are conditions in which the marrow produces excessive numbers of certain cells, but these cells are functionally impaired.
Viral Infections, Autoimmune Conditions, Toxins, and Drugs
Numerous acquired factors can injure bone marrow:
- Viral infections such as parvovirus B19, hepatitis viruses, and HIV.
- Autoimmune disorders that mistakenly attack hematopoietic cells.
- Toxins and chemicals, including benzene and pesticides.
- Medications like certain chemotherapies and antibiotics.
Complications of Untreated Marrow Failure
If untreated, marrow failure can lead to:
- Severe anemia, causing fatigue, shortness of breath, and heart strain.
- Life‑threatening infections due to low WBC counts.
- Severe bleeding from thrombocytopenia.
- Progression to leukemia in disorders like MDS.
Early intervention and expert care as provided by Kannappa Memorial Hospital’s hematology team significantly reduce these risks.
Symptoms and Signs
Clinical manifestations of marrow dysfunction relate directly to the deficient cell lines.
Fatigue, Pallor, and Anemia‑Related Symptoms
When RBCs are low:
- Persistent tiredness
- Pale skin and mucous membranes
- Dizziness or rapid heartbeat
Frequent Infections Due to Low WBC
A reduced white cell count leads to:
- Recurrent fevers
- Persistent or unusual infections
- Slow healing from minor wounds
Easy Bruising and Bleeding
Low platelets cause:
- Bruises with minimal trauma
- Nosebleeds or gum bleeding
- Heavy menstrual bleeding
Other Systemic Signs
Depending on the underlying disease, patients may also experience:
- Bone pain
- Weight loss
- Night sweats
- Enlarged liver or spleen
Accurate interpretation of these signs is a cornerstone of evaluation at comprehensive centers like Kannappa Memorial Hospital.
Types of Bone Marrow Diseases and Syndromes
Understanding specific marrow diseases helps tailor treatment:
- Leukemias: Leukemia is a cancer of blood‑forming tissues characterized by uncontrolled proliferation of abnormal white cells. Subtypes include acute and chronic leukemias, each with distinct clinical behavior.
- Aplastic Anemia: A severe form of marrow failure where all lineages are affected. It is a medical emergency if severe.
- Myelodysplastic Syndromes (MDS): MDS represents clonal marrow disorders where ineffective hematopoiesis results in cytopenias and progression risk to leukemia.
- Myeloproliferative Disorders (MPD): Conditions like polycythemia vera, essential thrombocythemia, and myelofibrosis fall into this category, involving overproduction of blood cells.
- Congenital / Inherited Syndromes: As described earlier, genetic defects such as Fanconi anemia and Diamond‑Blackfan anemia affect marrow from early life.
- Iron Deficiency and Other Nutritional Anemias: While not true marrow failure, deficiencies in iron, vitamin B12, and folate can greatly impair cell production and mimic marrow pathology.
- Rare Syndromes: Emerging genetic disorders like MIRAGE syndrome, DADA2 deficiency, and Pearson syndrome present with marrow failure accompanied by multisystem involvement and require genetic diagnostics.
Diagnosis
Accurate diagnosis relies on a multimodal approach.
Complete Blood Count (CBC)
A CBC is the first screening test, quantifying:
- Hemoglobin (Hb)
- White cell count
- Platelet count
Patterns of cytopenias suggest specific disease categories.
Bone Marrow Biopsy and Aspiration
This is the gold standard. It provides:
- Cellular architecture
- Morphological details
- Blast percentage
- Fibrosis evaluation
Genetic and Molecular Testing
Modern diagnostics include:
- Cytogenetics (chromosomal analysis)
- Mutation panels (e.g., FLT3, JAK2, TP53)
- Next‑generation sequencing
These tests improve classification and guide targeted therapies.
Imaging Studies
Ultrasound or CT scans may be necessary to look for organ enlargement or extramedullary disease.
Inherited vs. Acquired Differentiation
Clinicians use family history, age of onset, physical signs, and specialized tests to distinguish between inherited and acquired causes essential for prognosis and genetic counseling.
Treatment and Management
Treatment strategies depend on the specific diagnosis, severity, age, and patient preferences.
Supportive Care
Supportive therapies form the backbone of initial management:
- Blood transfusions
- Platelet support
- Antibiotics for infections
- Growth factors like G‑CSF to stimulate marrow
These measures stabilize patients and prevent emergency complications.
Immunosuppressive Therapy
In conditions like aplastic anemia, immune suppression (e.g., anti‑thymocyte globulin + cyclosporine) can allow recovery of hematopoiesis.
Bone Marrow / Stem Cell Transplantation
Hematopoietic stem cell transplant (HSCT) is a potentially curative option for:
- Severe aplastic anemia
- High‑risk MDS
- Leukemias
- Inherited marrow failure syndromes
At Kannappa Memorial Hospital, multidisciplinary transplant teams ensure careful donor selection, conditioning regimens, and long‑term care.
Disease‑Specific Treatments
- Leukemias: Chemotherapy, targeted agents (e.g., tyrosine kinase inhibitors), immunotherapies.
- MPDs: Agents to control blood counts, JAK inhibitors.
- MDS: Hypomethylating agents, targeted therapies.
Clinical Trials and Advanced Care Options
Clinical research constantly expands therapeutic options. Patients may benefit from novel agents, immunotherapies, and precision medicine approaches through carefully monitored trials.
Prognosis and Outlook
Prognosis varies widely:
Factors Affecting Survival / Recovery
- Disease type and stage
- Age and performance status
- Genetic mutations
- Response to therapy
- Access to transplant
For example, lower‑risk MDS often has a good outlook, whereas high‑risk leukemia may require aggressive therapy.
Potential Complications
Despite treatment advances, complications like infections, bleeding, graft‑versus‑host disease (post‑transplant), and secondary cancers may occur.
Long‑Term Monitoring
Survivors require regular follow‑ups, CBC checks, organ function tests, and preventive care.
Prevention and Lifestyle Considerations
While genetic bone marrow disorders cannot be prevented, certain strategies can help reduce the risk of acquired marrow problems:
Avoiding Known Toxins and Drugs
Limit exposure to substances that can harm bone marrow, such as:
- Benzene
- Tobacco smoke
- High‑dose radiation
- Certain medications without medical supervision
Genetic Counseling
For families with hereditary bone marrow disorders, counseling and prenatal diagnosis can help guide planning and early care.
Regular Monitoring
Individuals with high-risk exposures or a history of low blood counts should undergo periodic evaluations to detect early changes in bone marrow function.
Living with Bone Marrow Failure
Managing a bone marrow disorder or bone marrow failure requires a combination of medical care and practical lifestyle adaptations:
Self‑Care and Monitoring
- Track symptoms and blood counts regularly to detect changes early.
- Keep your vaccinations up to date to help lower the risk of infection.
- Maintain good nutrition and exercise safely within your limits to support overall health.
When to Seek Emergency Care
- High fever
- Heavy or unexplained bleeding
- Severe symptoms such as chest pain, difficulty breathing, or other serious signs.
Support Resources
Engaging with support groups, counseling services, and coordinated care teams can significantly improve quality of life and emotional wellbeing for those living with a bone marrow disorder.
Conclusion
Bone marrow failure and related disorders represent a spectrum of conditions that compromise blood cell production, leading to serious health issues. Early detection, accurate diagnosis, and prompt management are keys to better outcomes.Kannappa Memorial Hospital combine expert clinical care, advanced diagnostics, multidisciplinary treatment, and patient support to deliver comprehensive marrow disease management.
Whether through supportive care, cutting‑edge treatments, or life‑saving transplantation, individualized care helps patients live fuller, healthier lives despite marrow challenges.If you or a loved one is facing symptoms like fatigue, infections, or easy bruising, professional evaluation can make all the difference and specialized centers like Kannappa Memorial Hospital can offer the expertise needed for optimal care.